-
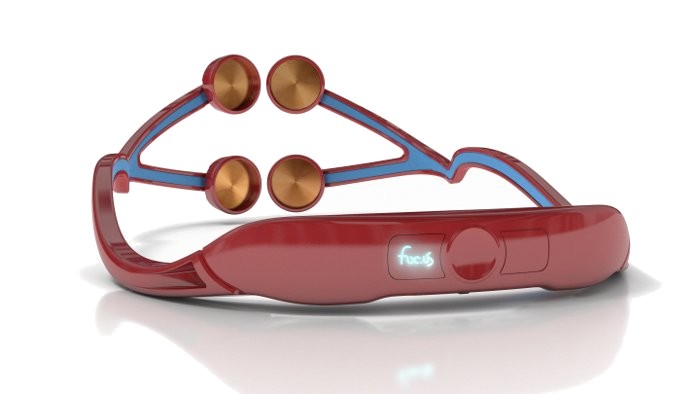
The Foc.us Has Landed!
Is foc.us FDA approved? No. The focus gamer headset offers no medical benefits, is not a medical device, and is not regulated by the FDA.
- When will my headset ship? If ordered today your headset should ship by the end of July 2013. Subsequent production orders are scheduled from October 2013.
- Will Android be supported? Yes, we are fully committed to providing Android support as soon as Android contains the required bluetooth 4.0 apis. We ♥ Android.
- Is the headset safe? The focus headset has been tested to all required regulatory standards including CE Safety standard EN60601-2-10: 2001 and EN60601-1: 2006.
- What does the headset do? The headset passes a small electric current <2.05mA through the prefrontal cortex of the wearer.
- Who should not use foc.us? The headset is not a toy, is not recommended for under 18s, epilepsy suffers or people with implants. It should not be used in the treatment of any medical conditions.
via FOC.US – transcranial direct current stimulation for gamers.