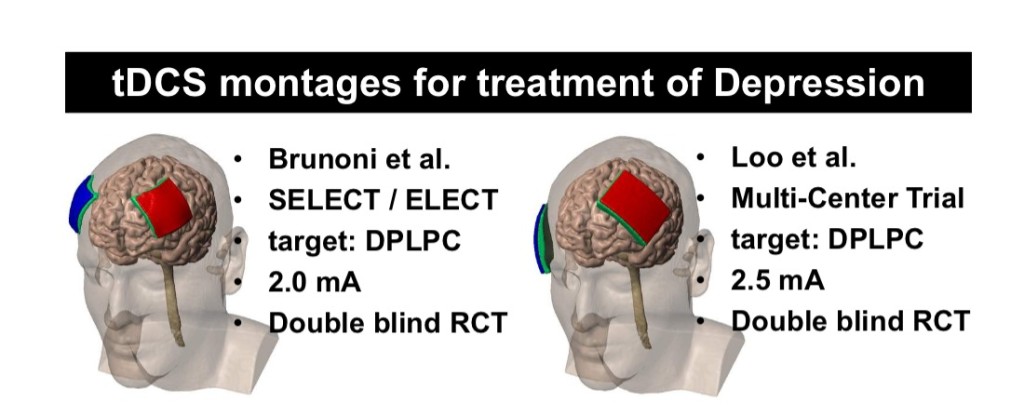
If you’ve been following along then most of what’s discussed in this May, 2018 lecture won’t surprise you, but it’s nicely put together and I would say, emphasizes the validation of applying stimulation while ‘doing something’ (Functional Targeting). The idea being that the stimulation then amplifies the activity of the neurons involved in the task. Though it does beg the question, What would be an appropriate task to involve a patient with in the treatment of depression? I think it’s important to note that most of what is discussed, in terms of any affects tDCS may have, remain hypothetical!
The 2019 joint meeting of Neuromodulation:The Science and NYC Neuromodulation is about to start and a page devoted to abstracts has been published. It will be updated as more info becomes available. Want a look into the future of neurostimulation? Here it is!
Dr. Brent Williams of SpeakWisdom recently published an article on the new LIFTiD tDCS device, How to Use the LIFTiD Headset for Depression Relief. The device (which is available at Caputron) has yet to be extensively reviewed but its main advantage, defined electrode placement, could also be seen as a weakness.