Honestly I just burned out on following along with a lot of science that didn’t seem to be getting anywhere. What attracted me to tDCS were the stories and studies that implied accelerated learning and improved memory skills were just around the corner. What kept me interested were depression studies that did seem to demonstrate effectiveness. And considering my own bias against pharmaceuticals, even if the positive effects of tDCS for depression were placebo, I considered it worth talking about and promoting. I still do, for depression.
But tDCS is fairly easy to work with and that means lots of young scientists doing low-powered studies, the upshot of which might be a published paper, but not much else.
What actually stopped me in my tracks was the sudden realization of how little we know about electricity in the human body.
It was this Sean Carroll podcast with Michael Levin where he discusses manipulating the growth of tadpoles and flatworms with electricity (among many other things related to bioelectricty) that made me question the safety and sensibility of introducing current into our brains! There are some profound first-level mysteries at play here, and until we know more, OR, there is a solid ‘do this – get that result’ application of tDCS, I’m taking a wait and see perspective.
Thanks for following along!
John
Category Archives: Paper
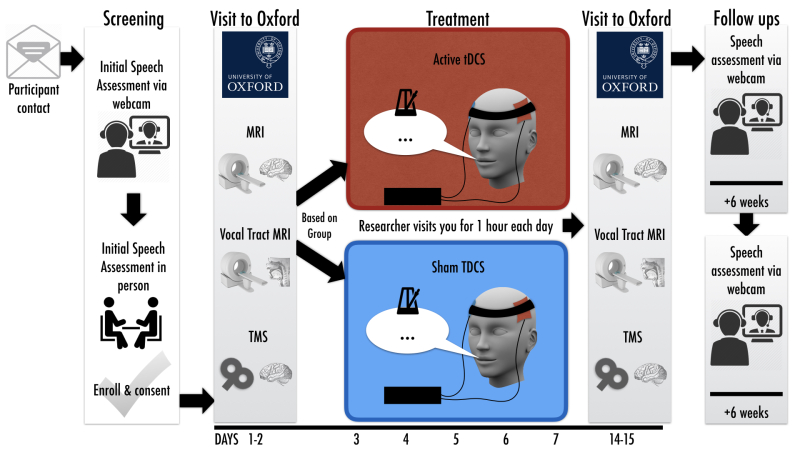
INSTEP Stutter / tDCS Study
Live anywhere near Oxford England and have a stutter? This study is currently recruiting.
More info: https://insteptrial.wordpress.com/
In this study, we want to see if a five-day course of tDCS combined with fluency training will improve speech fluency more than the fluency training on its own. We also want to see if such improvements are maintained over a period of three months.
Ed Boyden Synthetic Neurobiology
Tyler Cowen interviews Ed Boyden of MIT in this excellent interview. (Seriously, listen to this interview).
Ed Boyden is a neuroscientist and his lab, Synthetic Neurobiology Group is serving up a steady stream of cutting edge brain research. Especially of interest to DIYtDCS viewers is his involvement in a new form of non-invasive brain stimulation able to reach deep into the brain…
The method, called temporal interference, involves beaming different electric frequencies, too high for neurons to respond to, from electrodes on the skull’s surface. The team found that where the currents intersected inside the brain, the frequencies interfered with each other, essentially canceling out all but the difference between them and leaving a low-frequency current that neurons in that location responded to, Dr. Boyden said.
NEW YORK TIMES New Electrical Brain Stimulation Technique Shows Promise in Mice
Paper: Noninvasive Deep Brain Stimulation via Temporally Interfering Electric Fields
But what really caught my ear in this interview was making the connection that Dr. Boyden is working with MIT neuroscientist Li-Huei Tsai. You’ll recall Dr. Tsai from earlier posts that discuss her work around treating Alzheimer’s using Gamma waves. Gamma Waves Enhance the Brain’s Immune System to Treat Mice with Alzheimer’s disease It turns out that Boyden and Tsai are working on a commercial venture that will explore this research together. The company is called Cognito Therapeutics.

It’s still early days for Cognito, though they are recruiting for studies. I’ll be following along closely.
Gamma Waves Enhance the Brain’s Immune System to Treat Mice with Alzheimer’s disease.

[Update 3/18/19] Exciting news in the fight against Alzheimer’s!
Dr. Tsai has a new lab. Tsai Laboratory
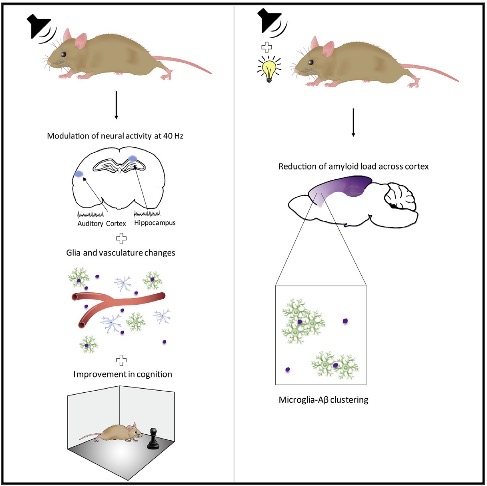
In the new study, researchers added acoustic stimulation that pulsed at the same frequency as the visual stimulation. And when mice — which were bred to develop Alzheimer’s-like symptoms — were exposed to both, researchers saw that the neurons in several of their brains’ key memory circuits chimed in and began humming along at exactly the same frequency.
LA Times https://www.latimes.com/science/sciencenow/la-sci-sn-alzheimers-brain-waves-20190315-story.html
What came next was remarkable and unexpected, even to the authors of the study.
In the wake of the sound and light sessions, an army of newly energized immune cells descended on several areas of the treated brains, including those most affected by dementia. Then they set to work with a vengeance on some neglected housecleaning.
A Possible Alzheimer’s Treatment With Clicks and Flashes? It Worked on Mice (NYT)
Light and sound stimulus therapy generates a buzz in Alzheimer’s research world (LAT)
——–
[Update 2/3/18] Nature reports on updates since the information about the research first emerged. How flashing lights and pink noise might banish Alzheimer’s, improve memory and more
——— Original article
I was first alerted to the story from a December 7 article in the Guardian, “Strobe lighting provides a flicker of hope in the fight against Alzheimer’s“. Researchers from the Picower Institute for Learning and Memory at MIT, working with (let’s call them) ‘Alzehiemer’s mice’, had discovered that flashing a light at 40hz (on-off at 40 times per second) increased gamma wave oscillations in the brain which led to the reduction of Amyloid beta (think, plaque) through the activation of microglia ‘clean-up’ immune cells. Here, let them explain it!
The paper, Gamma frequency entrainment attenuates amyloid load and modifies microglia makes clear that the light-flickering affected the visual cortex, which makes sense, as the light reaches the brain through the eyes. But wait, thinks I, what about tACS (transcranial Alternating Current Stimulation)… haven’t I seen numerous papers implying the ability to ‘entrain’ brain waves with tACS? What if you could increase 40hz Gamma in other parts of the brain? (Google Scholar Search: transcranial alternating, entrain, gamma)
But then I discovered that Radiolab just covered this exact story and it’s totally amazing! Really a must listen. So fun to hear the researcher’s amazement at this accidental (sort of) discovery!
So what’s with the photo of the Foc.us v2 device set up for a 40hz tACS session? Just that…

More about The Picower Institute for Learning and Memory at MIT
Physical Medicine and Rehabilitation Non-Invasive Brain Stimulation (NIBS) | John Hopkins
Michael was told he had reached his limit on what he could do. This was not something Michael and his partner, Jason, would accept. They began to look for other options and found Johns Hopkins Physical Medicine and Rehabilitation’s Non-Invasive Brain Stimulation (NIBS) as Michael’s answer to improve. Watch to learn more about Pablo Celnik and his team’s approach to setting Michael on a path to recovery.
John Hopkins Physical Medicine and Rehabilitation
Doc Johnson’s DIY Build | diligent5.org
From a comment to the blog. Doc Johnson shares this beautiful DIY tDCS build. Thanks Doc!
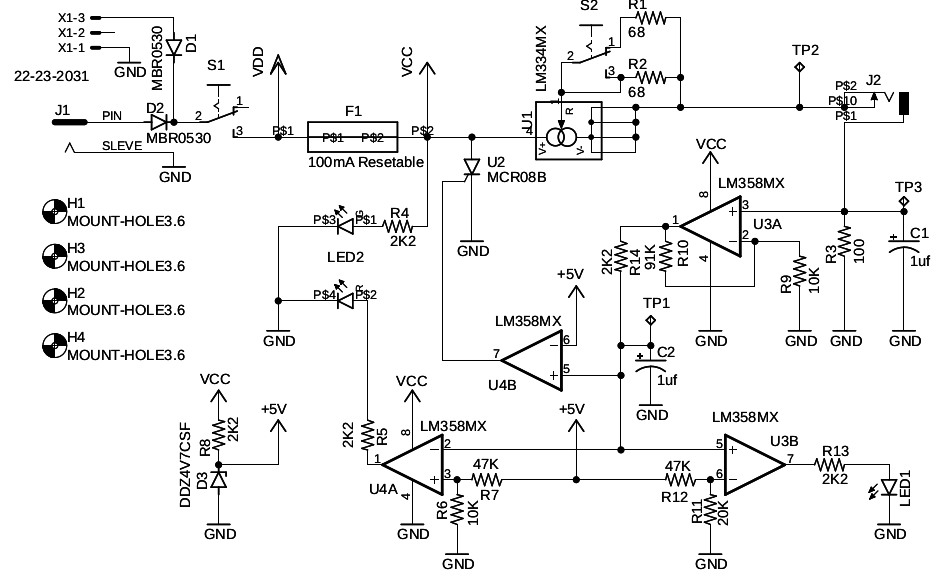
I’m personally just beginning experimenting with TDCS, and as an avid DIYer, I built my own. The open designs I’ve seen seem to be a little short on features (particularly a fail-safe over-current protection), so I decided to use my background as an Electrical Engineer to start from scratch. It might be useful for others who are interested in building their own.
The design incorporates a feature that trips something equivalent to a circuit breaker if more than about 4.8mA of current flows through the electrodes. It should never happen based on the current regulator design, but I’ve seen too many pieces of silicon short out in a way that would be dangerous in this situation. I finished testing the design this weekend, and am quite happy with it. All told, the parts are about $20, not counting the hours spent designing, building, testing, redesigning…
The design and links for how I built the circuit board are located at https://www.diligent5.org/?p=1993 I built it using surface mount components because I like the tighter resistor tolerances available, but theoretically the components (or something equivalent) are available as through-hole components for the less adventurous. Cheers, Doc Johnson
- TDCS Controller Step 2 – Building The Circuit Board
- TDCS Controller Step 3 – Testing the Design
- TDCS Controller Step 4 – Redesign
- TDCS Controller Step 5 – Making Electrodes

tDCS and Parkinson’s
First of all I need to reiterate that I am not a health professional by any means (I’m a retired musician). I was attracted to tDCS when cognitive enhancement effects were first reported back in 2012. Now years later I’m still not convinced there’s anything more than placebo going on with tDCS as far as cognitive enhancement goes, though as I’ve stated elsewhere on the blog, the research seems more conclusive around tDCS being an effective treatment for depression.
Parkinson’s is a progressive, debilitating, and at present, incurable disease that robs people of a wide variety of function, but especially movement and speech-related abilities. I have a close friend with Parkinson’s so I’ve been following the related tDCS literature. A recent study suggests that tDCS may prove to be an effective way to help with movement issues.
In Effects of Transcranial Direct Current Stimulation (tDCS) Over the Frontal Polar Area on Motor and Executive Functions in Parkinson’s Disease; A Pilot Study researchers at Toyama University Hospital, Japan, found that anodal tDCS improved motor function in patients with Parkinson’s. Note that this was a very small study (9 patients, early in their Parkinson’s). In this blog post I will attempt to present a DIY version of the study protocol.
To the best of our knowledge, this is the first clinical research reporting that tDCS over the FPA successfully improved the motor and non-motor functions in PD patients. These findings suggest that tDCS over the FPA might be a useful alternative for the treatment of PD patients.
The first thing I noticed was that the montage (where to put the electrodes), “tDCS over the frontal polar area (FPA)” was unusual, therefore not easily verified by looking at other studies. The researchers had previously determined that stimulation of the FPA in healthy subjects improved hand dexterity (Cerebral functional imaging using near-infrared spectroscopy during repeated performances of motor rehabilitation tasks tested on healthy subjects). From the paper (my edits, for brevity and clarity in regards my purpose here)…
The stimulation current was delivered … through a pair of saline-soaked sponge electrodes (5 × 7 cm) over the frontal polar area FPA, and occipital area (OPA).
tDCS was applied for 1 week (5 times/week: Monday–Friday).
A constant current of 1.0 mA was delivered for 900 seconds (15 minutes).
During the tDCS protocol, the patients received traditional physical therapy in the upper extremities (stretching and muscle strength exercise) while sitting in a chair.
According to the 10-20 EEG system (pdf), FPA is defined as Fp1 and Fp2, and OPA is positioned near O1 and O2. Digging into the previous study I determined that they used FPZ to target FPA, and OZ to target OPA.
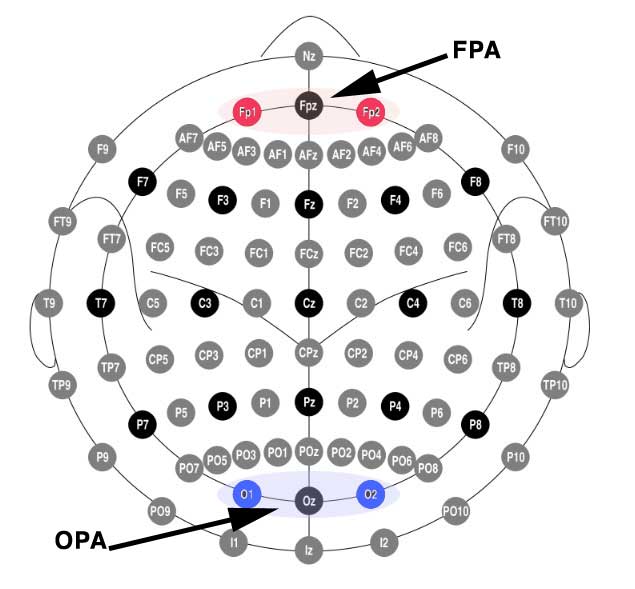
Yes but… what does that look like on a head? Here I’m indicating the electrode targets using the Foc.us placements head model.
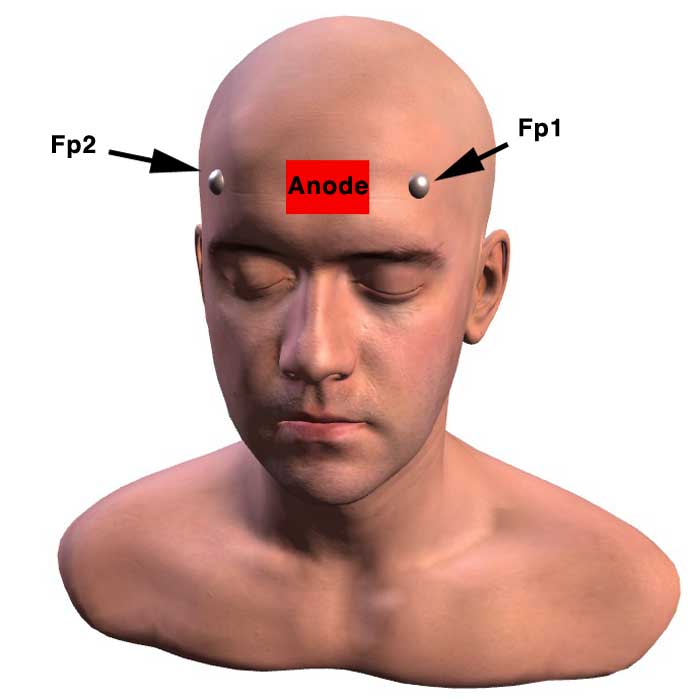
(Above) Anode placed at Fpz to target the frontal polar area (FPA)
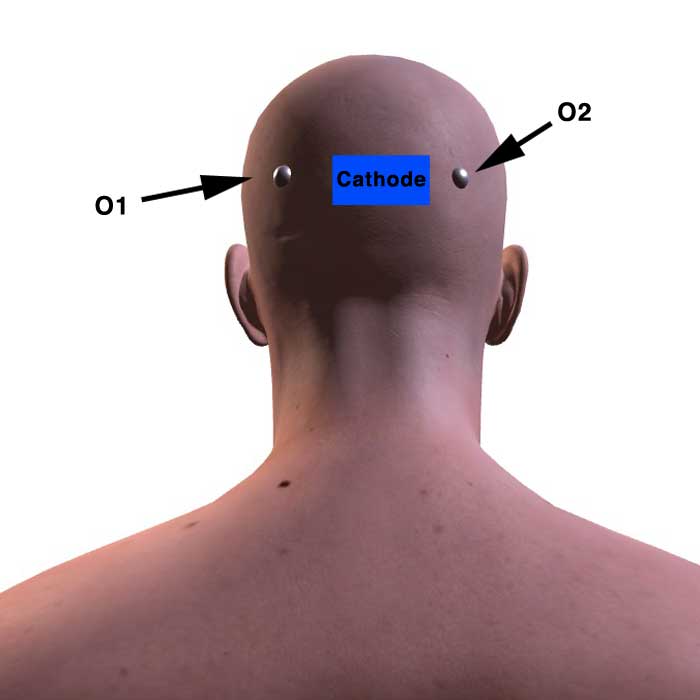
(Above) Cathode placed at Oz to target occipital area (OPA).
So basically, there you have it…
1 mA stimulation, (approx) 2″ x 2″ saline-soaked sponges, for 15 minutes placed at Anode-Fpz and Cathode-Oz, every day for five days. Note that the study participants underwent their usual regimen of physical therapy while the tDCS was being applied.
Am I suggesting you try this? No, I’m not.
I personally do intend to see if this can help my friend whose walking and talking are becoming difficult. For this reason I wanted to be as clear in my understanding of the study as possible.
Depression treatment technique uses new helmet therapy | BBC News
Updated 9/3/18: TMSuino3 A low-cost arduino-based TMS (transcranial magnetic stimulation) device. 
This github project is a step-by-step manual. Anyone with a high school diploma should be able to follow it and build a TMSuino themselves. That’s what I was aiming for. TMSuino’s principle of operation is taken from a scientific paper published in the 1990s. So it is free of valid patent claims. Costs for parts, materials and shipping should be around 150,- dollars/euros. There is no soldering required! Estimated build time is 2 1/2 hours.
A comment on the blog from Martin Mueller informed me of this very interesting new arduino-based TMS device. I’ve also posted a link to the r/diytdcs Reddit page in hopes of hearing feedback from more technically-minded folks. I will update this post as info emerges. TMSuino3 seems to be loosely based on the work of Steen Dissing whose device we looked at in 2015 (see below) where it’s referred to as Transcranial Pulsating Electro Magnetic Fields therapy or t-PEMF.

The Introduction to the github article collects the author’s frustrations and suspicions around transparency in the bipolar research community and basically comes away from the experience hypothesizing what sounds like a conspiracy. Wait, big pharma and medical device companies manipulating the market to ensure maximum profit? Either way, he shares an article that confirms Steen Dissing’s frustration in getting his device accepted and adopted for treatment of depression in Denmark. An Inventor’s Triumph and Frustration.
It turns out a medical grade device has been developed and approved for treatment of depression in Denmark. The Re5, made by Navamedic. There are a collection of research papers linked to at their site.
 ————————– Original 2015 article.
————————– Original 2015 article.
Thanks to reader Jerico for alerting me to this. Transcranial Pulsating Electro Magnetic Fields therapy is new to me. It does not seem to be experiencing anywhere near the level of research activity that is going on around tDCS. Some of the research I’m finding dates from 2001 (though the BBC article the quote is from and linked to below is from 2014). But just to have it on our radar, and because the helmet looks so cool…
“The helmet is amazing,” said Annemette Ovlisen, a graphic artist who suffered recurrent depression for 16 years and a participant in the Hillerod trials.
It’s like the fog lifts. It was like somebody hit the reset button.”The device contains seven coils that deliver a dose of Transcranial Pulsating Electro Magnetic Fields (T-PEMF) to brain tissues.The pulses are so minute that the patient cannot detect any sensation, and the only side effect so far is occasional “tiny” nausea that immediately disappears after treatment.
Prof Steen Dissing, of Copenhagen’s Faculty of Health Sciences is the helmet’s principal architect.
He said: “The device mimics electrical fields in the brain, and triggers the body’s own healing mechanism.”
The pulses activate capillaries in the brain, which form new blood vessels and secrete growth hormones.

I’ts like somebody hit the reset button and I was back to normal.

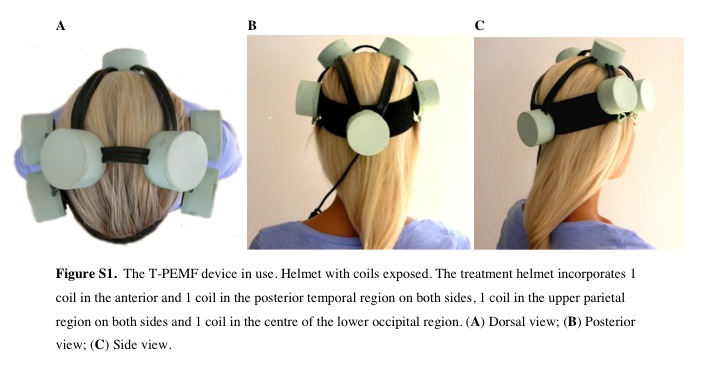 via BBC News – Depression treatment technique uses new helmet therapy.
via BBC News – Depression treatment technique uses new helmet therapy.
Understanding Noninvasive Brain Stimulation with Dr Marom Bikson | NeuroGal
Intro to, basics of tDCS.
Understanding Noninvasive Brain Stimulation with Dr Marom Bikson NeuroGal
Your Brain, But Better: Neurostimulation – Dr. Daniel Chao #488 | Bulletproof Radio
Dave Asprey and Dr. Daniel Chao, Co-Founder and CEO of Halo Neuroscience go deep into the past, present and future of neurostimulation.
Your Brain, But Better: Neurostimulation – Dr. Daniel Chao #488
The Research Institute of the McGill University Health Centre, Montreal, Testing TMS For Alzheimer’s
It ‘makes sense’ that stimulating neurons in areas of the brain affected by Alzheimer’s would be of benefit, but as the researchers state, we won’t know for sure until the science is complete. In the meantime, if you know anyone in Montreal with Alzheimer’s issues, the study is recruiting.
The study is still accepting new patients.To find out more, contact researchers at the Research Institute of the MUHC at 514-934-1934 ext 34439 or Rishanthi.sivakumaran [@] rimuhc.ca
Caputron Now Carrying Foc.us V2 Device
Robin at Caputron dropped me a note to let me know they are now carrying the Foc.us V2 device. Purchased alone, it does not include electrodes, but there is an option to add their ‘starter kit’ which includes the Caputron Universal Strap, Caputron Banana Adapter Cable for Focus Device, and Choice of 2×2 or 3×3 Electrodes. (Use diytdcs at checkout for generous discount). Foc.us V2 Device at Caputron.
If what attracted you to tDCS is all the news (and hype) around the possible benefits, cognitive and otherwise, that tDCS may provide, then I recommend the Foc.us V2 device. It’s had a thorough going over, and apart from the (then included) electrodes, proved to be an amazing piece of gear. tDCS, tACS, tRNS, tPCS in a single sub $300 unit with a software interface!
- Foc.us V2
- Universal Strap
- Banana Plug for Foc.us
- Caputron Electrode
Elsewhere on the blog I’ve stated that I recommend the ActivaDose ll device. This is an FDA approved device – it’s NOT FDA approved for tDCS – it’s approval is for use as an Iontophoresis device. The point is that the electronics and workmanship have attained an FDA level of approval. It’s simple and straightforward to use.
The only reason I haven’t recommended other tDCS devices on the market is because I’m not in a position to analyze the quality of their workmanship myself. I recommend the Activadose ll because people looking to experiment with tDCS for the treatment of depression can’t be assumed to have a toolset for determining the mechanical workmanship of an electrical device they’re going to be attaching to their heads! The Activadose ll, an FDA approved device, at least assures the buyer the device itself is of high quality. It’s also more likely to retain some resale value in the event someone decides later on to sell it.
I recommend the Foc.us V2 because of it’s variety of stimulation modes. Folks who are sophisticated enough about neurostimulation to be experimenting with cognitive enhancement would obviously benefit from having the option to test other forms of stimulation that frequently come up in the scientific literature.
When you use code diytdcs at checkout at Caputron you get a discount, and I get a small commission.
A Dancer, Musician, Jogger & Handball Player Try Halo Sport | Mario Marzo
I do hope to understand this better. Is it just that he was so impressed with his own Halo Sport experience that he was motivated to tell the world about it? Is it that Mario is a YouTube content creator and knew this would be compelling content? I will update the post as I learn more.
And here is the video Mario made in November, 2016 where he describes the impact using Halo Sport had on his piano playing.
Hey Mario, If you’re reading this drop me a line, I’d like to talk to you.
TMS For Depression Making Inroads Into Mainstream
Insurers are starting to cover TMS for depression (after determining that SSRIs or other medications aren’t working for the patient). A full course, 24-36 treatments, of TMS can cost well over $10k. Though this is purely conjecture on my part, one way tDCS might make it into the mainstream is as a method to ‘top up’ post-TMS treatment as effects begin to fade.
Published on Jun 19, 2017 | YouTube UCLA
As the number of people suffering from depression rises, doctors are looking for new, more targeted ways to treat it. One approach used by doctors at UCLA and a handful of other centers nationwide is to beam magnetic pulses deep into patients’ brains, a therapy known as transcranial magnetic stimulation (TMS). The therapy is time-consuming, and only a few hospitals or clinics offer it, but its ability to work in a fundamentally different way from medications is also what makes it so promising for people not helped by drugs.
Closer look at transcranial direct current stimulation | WPBF News
Features a variety of Foc.us devices. The doctor interviewed is Dr. Brett Osborn.