Update 2/2/17. Focus just announced functional Near-InfraRed Spectroscopy (fNIRS) capabilities for their EEG Dev kit! 
This was announced a few days ago and to be honest, I wasn’t sure what to make of it… a battery-looking EEG thing. Certainly I’m not a ‘Dev’ and so I left it to those who are to parse the details, still… Ah, yes, further details arrived today via email I’m happy to share with you (below). I do get the feeling this will make EEG devs excited.
Update 1/19/17 The focus site now has a photo of their new dry EEG electrode.
Thanks for all your feedback and questions about the focus EEG. A common question has been what exactly is included (see below) and is it everything required (yes).
Included in EEG Dev Kit
- foc.us EEG 24-bit 8-channel EEG with tES & Wi-Fi
- 8 active dry electrodes for EEG, plus bias & reference electrodes
- 2 active bio-potential electrodes for ECG, EOG, EMG or EKG
- 2 wet tES electrodes for tDCS, tACS, tPCS or tRNS
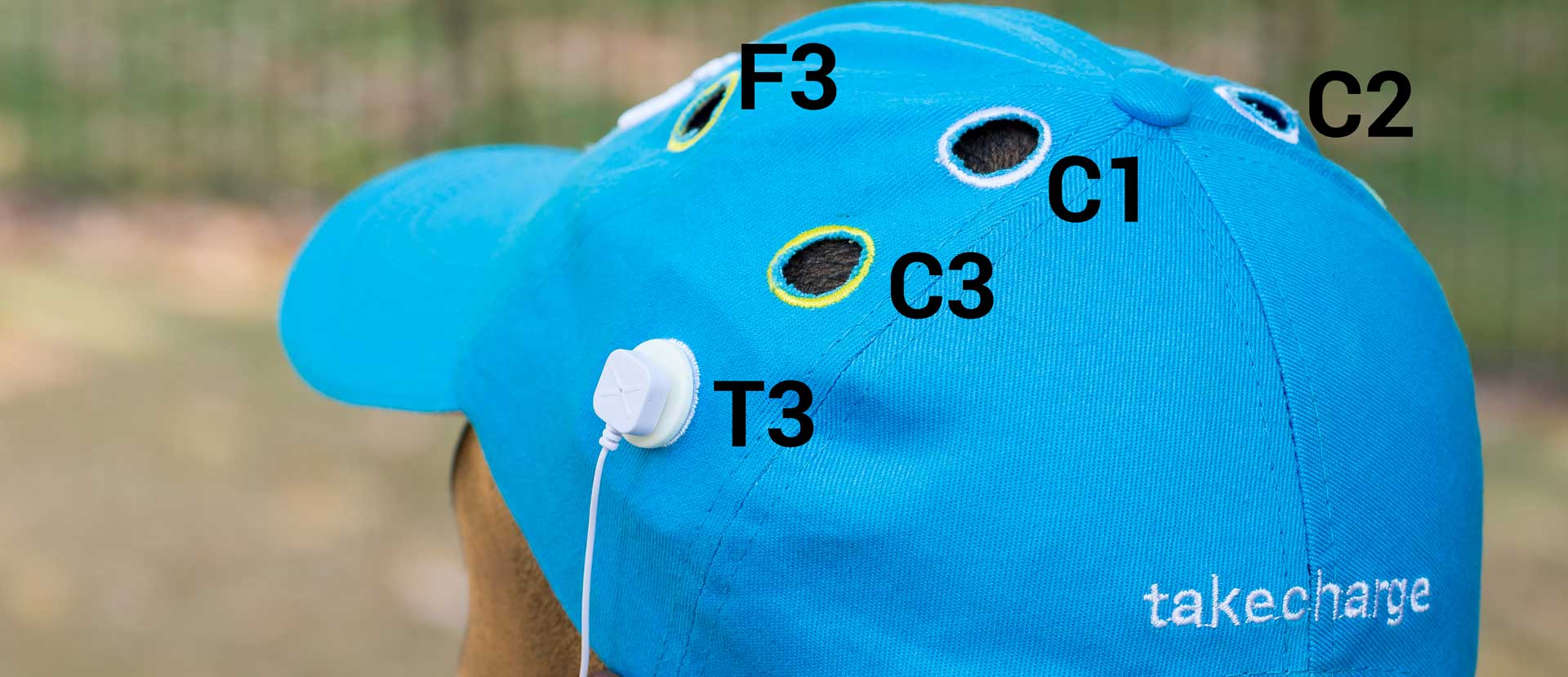
- 10-20 placement cap
- Mains power adapter for recharging
- Raw data access
Next week we will provide more details on the software and SDK for EEG processing.
Sincerely,
team focusP.S. The first 100 66 are available at only $999 $499 – half price!
What I’m excited about is the Focus EEG headset, but a recent tweet exchange indicates we’re a good year away from release.